Artificial Photosynthesis
A sustainable production method for fuel and value-added products inspired by nature
Photosynthetic core with chlorophyll for capturing sunlight and a catalyst (Mn) that triggers splitting of water molecule.
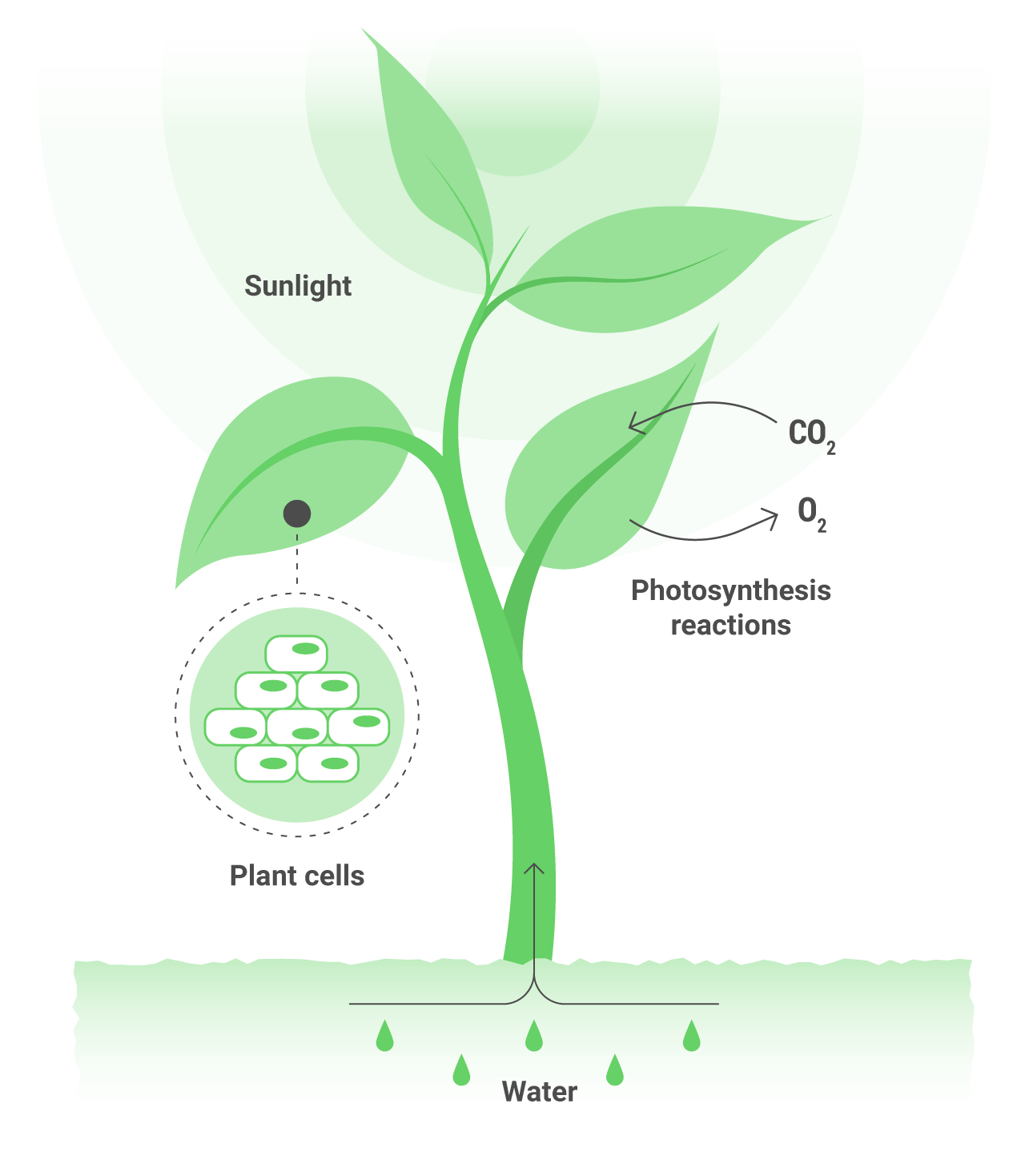
As a result, plants obtain the energy and nutrients necessary to live.
Material able to generate electrons from light and a catalyst to split water.
Mimicking the natural process is a way to get a renewable energy source. Sustainable fuel with a carbon-neutral footprint since it needs CO2 for its production.
A perfect model
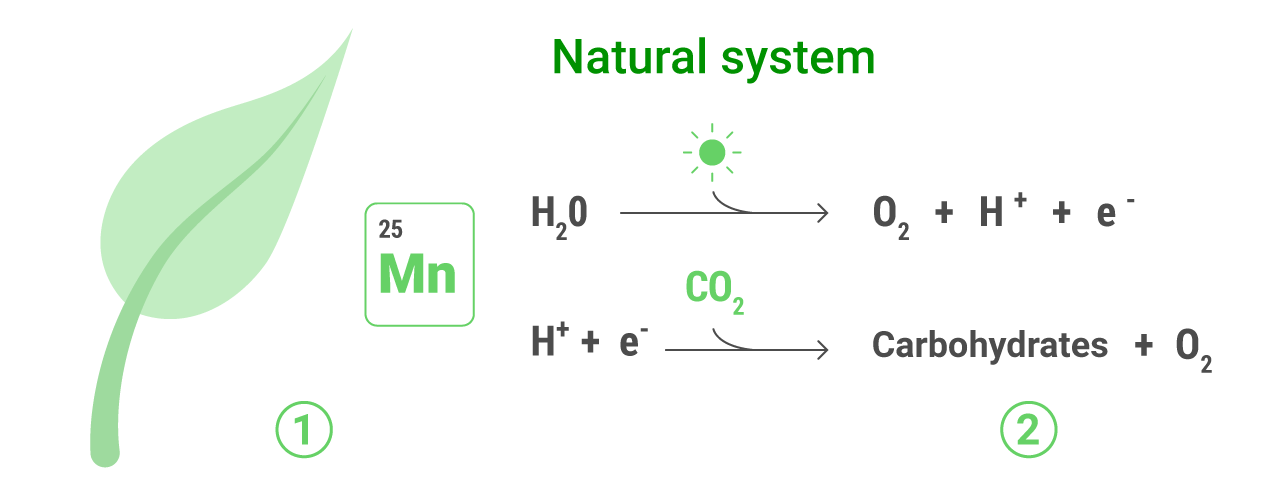
Plants live by using sunlight to trigger a chemical reaction that creates sugars form water and CO2. This process is called photosynthesis and it wouldn’t be possible without chlorophyll, which captures the sunlight, and a collection of proteins and enzymes that use it to break down H2O molecules into hydrogen (protons), electrons and oxygen. The electrons and hydrogen are then used to turn CO2 into carbohydrates, and the oxygen is expelled.
For these process to work, it’s essential the presence of catalysts, which lower the energy needed to carry out the different reactions. In plants, this function is carried out by manganese which is located within the photosynthetic core. A single atom of manganese triggers the natural process that uses sunlight to split water. Using manganese in an artificial system is a biomimetic approach – it directly mimics the biology found in plants.
Mimicking nature
Artificial photosynthesis is a biomimetic approach – it directly simulates the biology found in plants. In order to recreate it, an energy conversion system must be able to do two crucial things: harvest sunlight and split water molecules. All within a structure (probably built of nanotubes) that acts as the structural “leaf”.
From a scientific point of view, artificial photosynthesis can be divided into two reactions: first, the split of the water molecule, releasing oxygen and protons – that can be used to produce hydrogen, which in turn can be used as a fuel. Second, the conversion of CO2 into other organic molecules, that can also be transformed into fuels.
Unlike most methods of generating alternative energy, artificial photosynthesis has the potential to produce more than one type of fuel: hydrogen, methanol, formic acid, etc., depending on the catalyst used and the conditions of the “cell”.
A sustainable solution
Artificial photosynthesis allows us to obtain fuels from renewable sources like water, carbon dioxide, and sunlight. Besides, using CO2 in the production it turns it into an energy storage solution that it’s carbon neutral.
What are ICIQ
research groups doing?
ICIQ researchers are using different approaches to study artificial photosynthesis: from developing photocatalysts able to perform both photosynthetic reactions, to using computational methods to understand the inner workings of the reaction.
The Romero research group studies in detail the design principles that lead to efficient solar-energy collection, transfer and conversion in natural photosynthesis. Their goal is to transfer this knowledge into engineered bio-inspired chromophore-protein assemblies able to convert solar to electrochemical energy.
ICIQ’s computational groups are also delving into the mechanisms behind photosynthesis. The Bo group applies computational chemistry methods to determine catalytic reaction mechanisms, such as the transformation of the hydrogens found in water to dihydrogen – among other topics.
The López group is using computational methods to carry out “Theoretical Heterogeneous Catalysis,” say systematically searching for new catalysts to relevant chemical processes – among which are the transformation of water and CO2 into fuels.
The Galán-Mascarós group develops materials by combing their physical and chemical properties to further their energetic applications. The group is working in mimics of the natural photosystem, an inorganic cluster responsible for the catalysis of the transformation of water into protons and oxygen.
The Lloret-Fillol group works on the production of solar fuels by light-driven catalytic transformations of organic molecules. From a conceptual point of view, the group seeks to transfer concepts from artificial photosynthesis to the organic synthesis, to carry out transformations using sun-light as a driving force.
The Llobet group studies the catalytic oxidation of water to molecular dioxygen, given the implications of this reaction for new energy conversion schemes based on artificial photosynthesis. The group’s final objective consists in the photo-production of hydrogen from water and sunlight.