Solar Cells
A renewable source of energy that transforms light into electricity
The photons of light knock electrons free from atoms, which move to the p-type layer and left “holes” behind that will be filled by another electron.
When the n-type and p-type layers are connected with a metallic wire, the electrons of the p-type layer go back of the n-type layer. Electrons are moving from one layer to another, occupying and releasing holes, which generates a flow of electricity
A renewable source of energy
Solar or photovoltaic cells are devices that transform light into electricity. To do so, they use layers of different materials, with a central material (a semi-conductor) that absorbs light and starts a physical-chemical process to produce electricity.
Looking inside a solar cell
Solar or photovoltaic cells consists of layers of different materials, with a central one, a semi-conductor material, that absorbs light and starts a physical-chemical process to produce electricity.
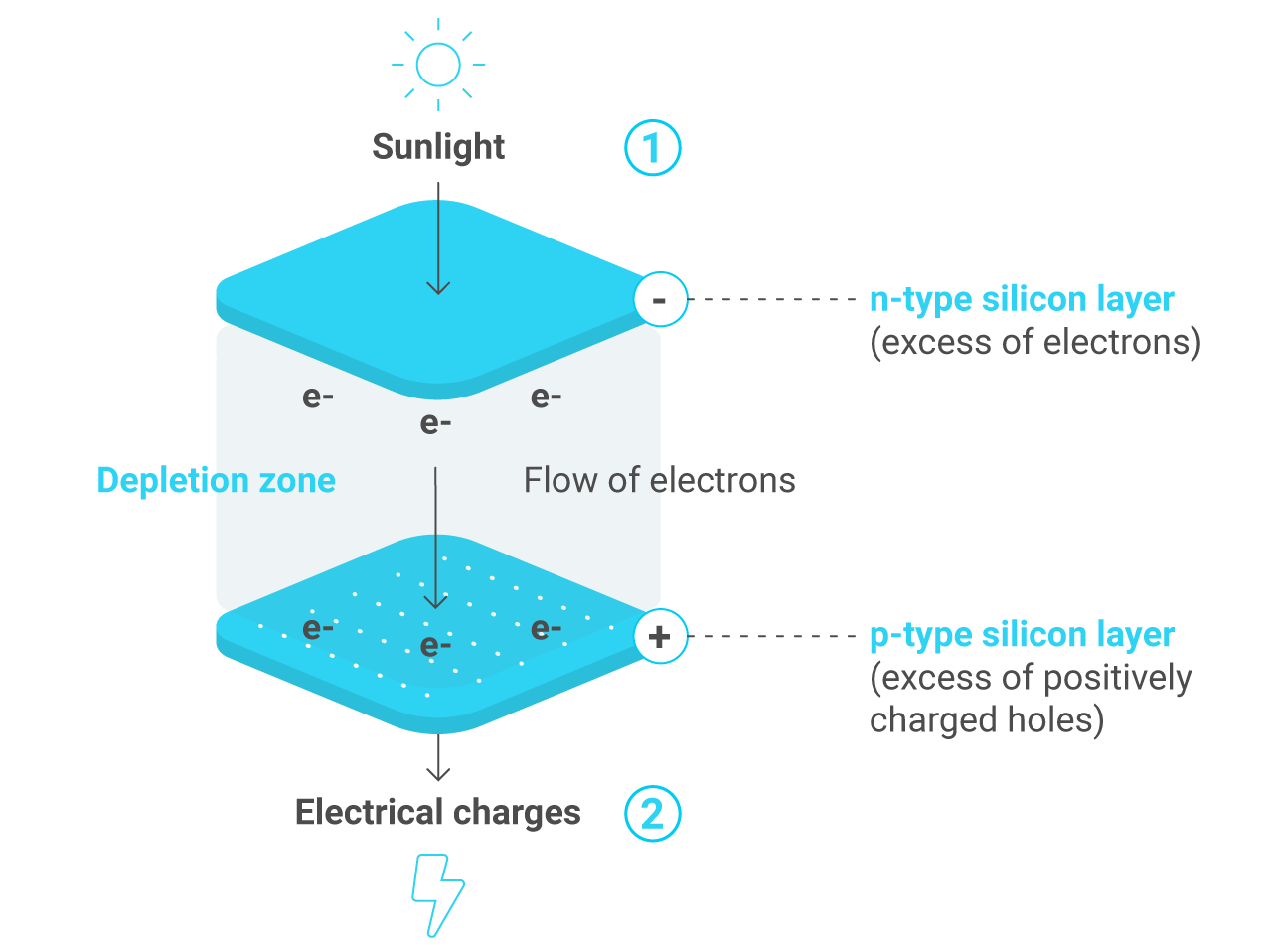
The structure of a solar cell is made up of a layer of n-type silicon placed next to a layer of p-type silicon. In the n-type layer, there is an excess of electrons, and in the p-type layer, there is an excess of positively charged holes (which are vacancies due to the lack of valence electrons). Near the junction of the two layers, the electrons on one side of the junction (n-type layer) move into the holes on the other side of the junction (p-type layer). This creates an area around the junction, called the depletion zone, in which the electrons fill the holes.
When all the holes are filled with electrons in the depletion zone, the p-type side of the depletion zone (where holes were initially present) now contains negatively charged ions, and the n-type side of the depletion zone (where electrons were present) now contains positively charged ions. The presence of these oppositely charged ions creates an internal electric field that prevents electrons in the n-type layer to fill holes in the p-type layer.
When sunlight strikes a solar cell, electrons in the silicon are ejected, which results in the formation of “holes”— the vacancies left behind by the escaping electrons —. If this happens in the electric field, the field will move electrons to the n-type layer and holes to the p-type layer. If you connect the n-type and p-type layers with a metallic wire, the electrons will travel from the n-type layer to the p-type layer by crossing the depletion zone and then go through the external wire back of the n-type layer, creating a flow of electricity.
Room for improvement
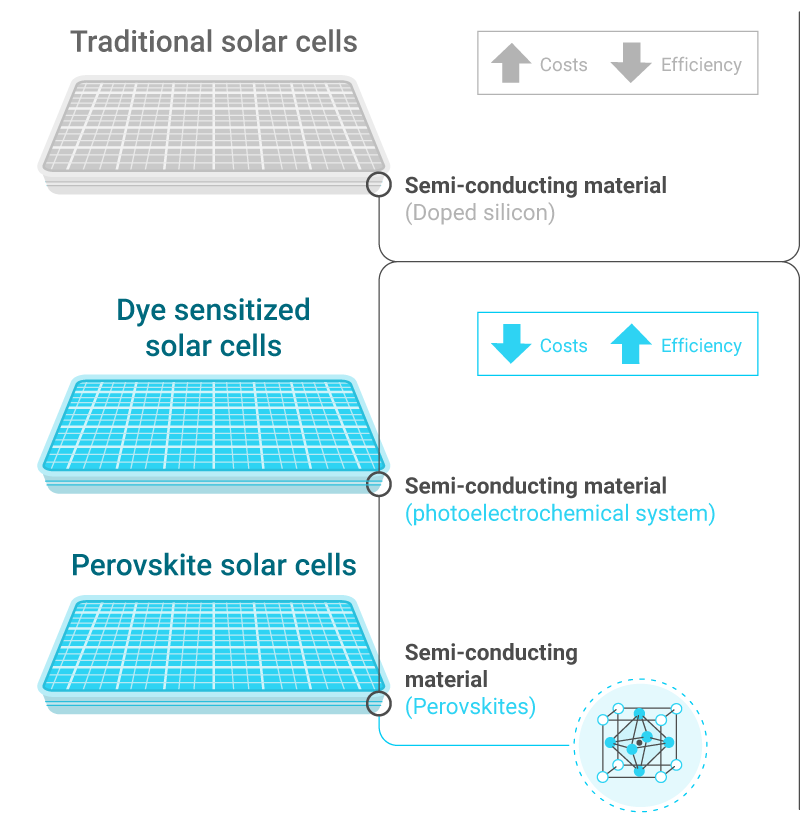
Traditionally, the central layer of a solar cells was doped silicon, but new research is putting forward new kinds of materials, peroviskites are an example. In recent years, scientists are investigating how to use them to build cheaper and more efficient solar panels. This class of materials were discovered in Russia in the 18th century, they are cheap, abundant and great at absorbing and emitting light. Peroviskites have the potential to change the dynamics and economics of solar power.
Perovskite solar cells are a type of solar cells which include a perovskite structured compound, most commonly a hybrid organic-inorganic lead or tin halide-based material, as the light-harvesting active layer. Perovskite materials are cheap to produce and simple to manufacture. Perovskite solar cells are therefore currently the fastest-advancing solar technology. With the potential of achieving high efficiencies and very low production costs, perovskite solar cells have become commercially attractive.
Dye sensitized solar cells are low-cost solar cells belonging to the group of thin film solar cells. They are based on semiconductors formed between a photo-sensitized anode and an electrolyte, a photoelectrochemical system. These cells are simple to make using conventional roll-printing techniques, is semi-flexible and semi-transparent which offers a variety of uses not applicable to glass-based systems, and most of the materials used are low-cost.
What are ICIQ
research groups doing?
ICIQ’s Palomares research group is developing perovskite-based solar cells using earth-abundant elements. The group also delves into the study of the charge transfer reactions within the solar cell’s layers, to better understand its inner workings, that limit the efficiency of current solar cells.
We design plant-inspired systems that allow you to absorb and transform sunlight into chemical energy more efficiently.